Performance. Apposition. Diversion.
Three advantages woven together.
Looking for every flow diversion advantage possible? Make the move from 48 to 64 wires with Surpass Evolve Flow Diverter—for the performance, apposition and diversion you’ve been asking for.

Wired for performance
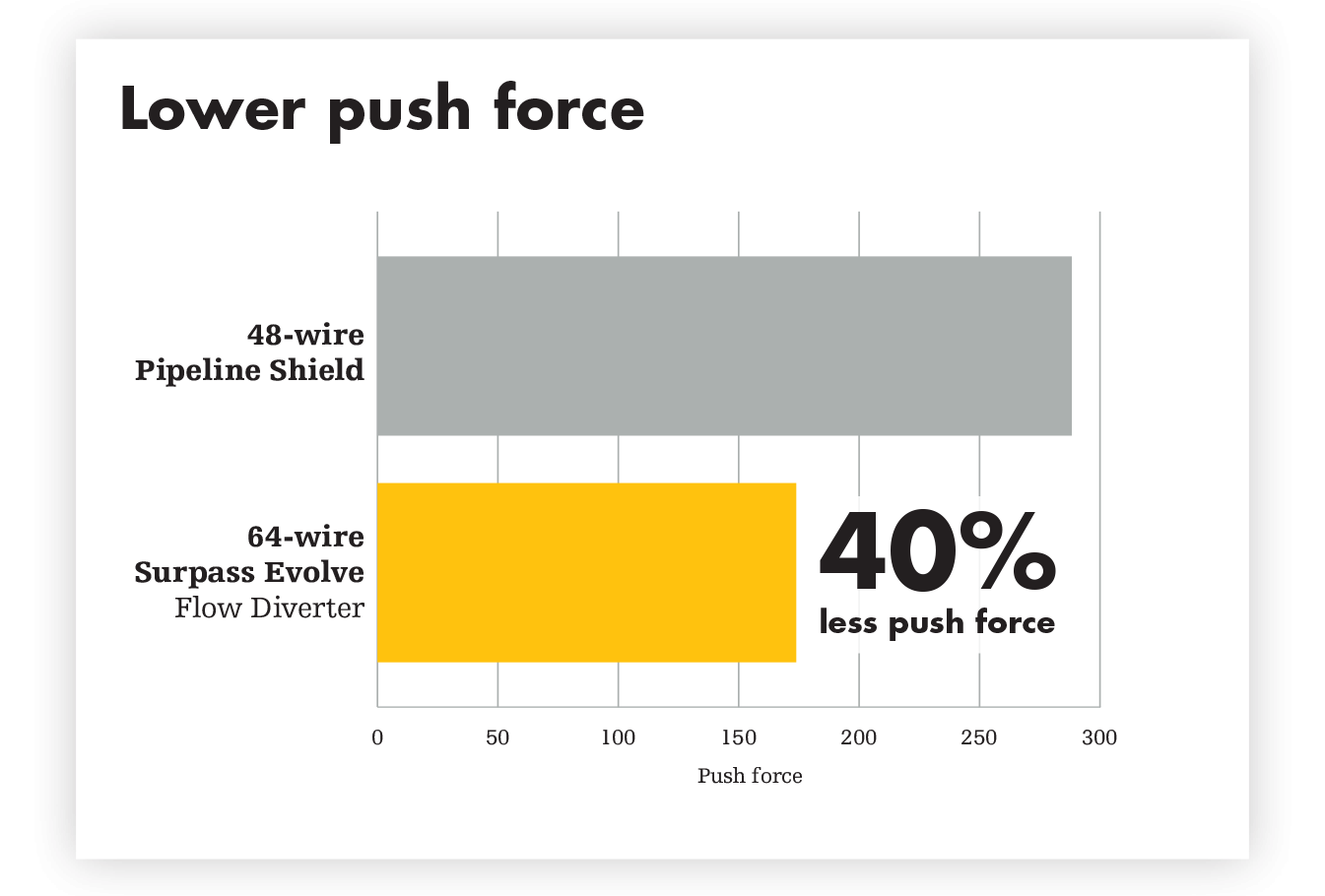
Designed for responsive navigation, precise distal landing and smoother resheathing. Unsurpassed performance happens here. The solid core delivery wire delivers 1:1 pushability and navigation through tortuous anatomy. The smaller petals can enable a faster opening and precise distal landing. And the innovative resheath pad makes for smoother resheathing and precise control.
Testing performed by Stryker. Data on file at Stryker.
Bench test results may not necessarily be indicative of clinical performance.
60% shorter procedure times with Surpass Evolve Flow Diverter vs 48-wire Pipeline1
The flexibility profile of the solid core wire is designed to match anatomy and partner with Excelsior XT-27 Microcatheter Standard Straight, for lower friction forces, even in highly tortuous vessels.
Wired for apposition
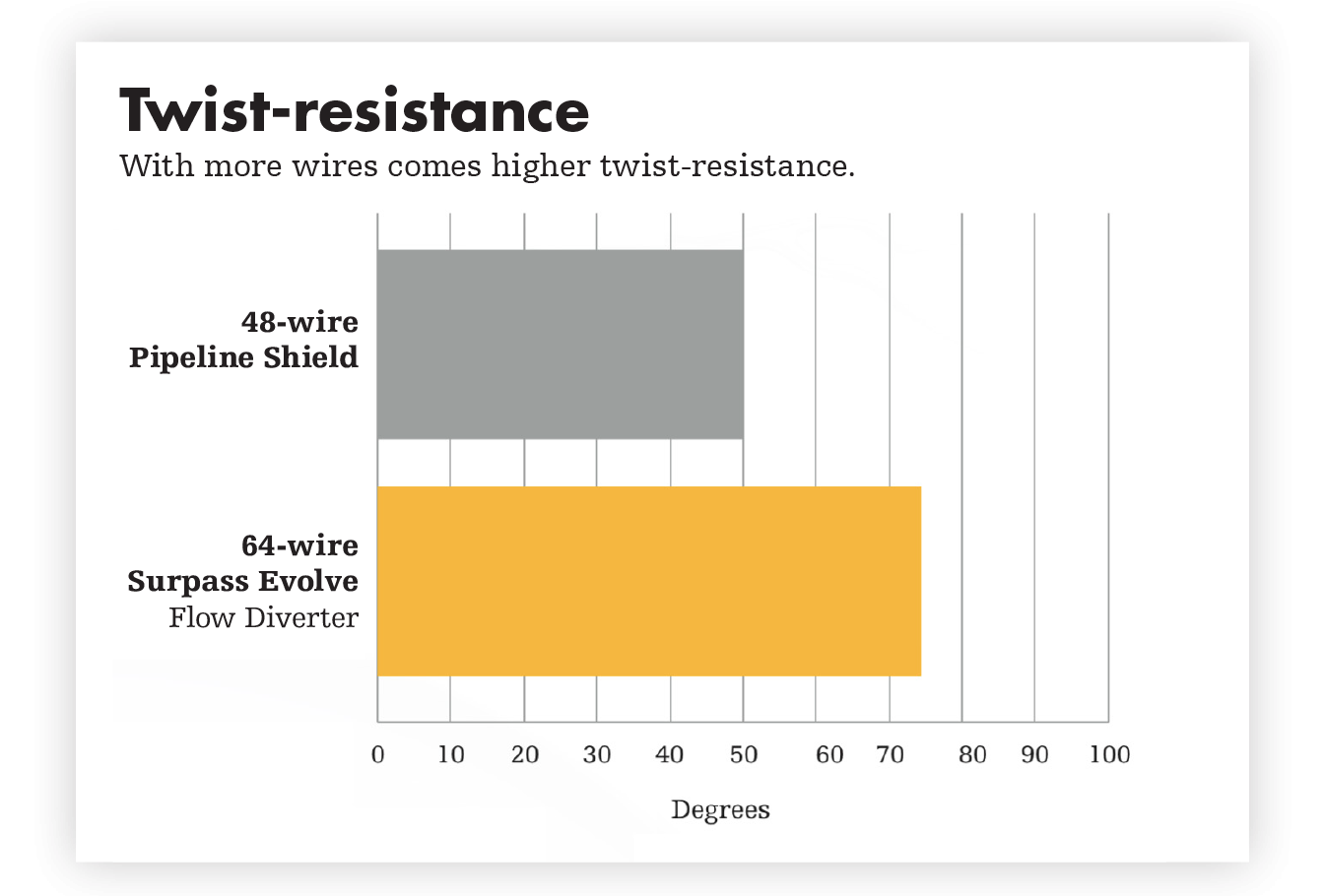
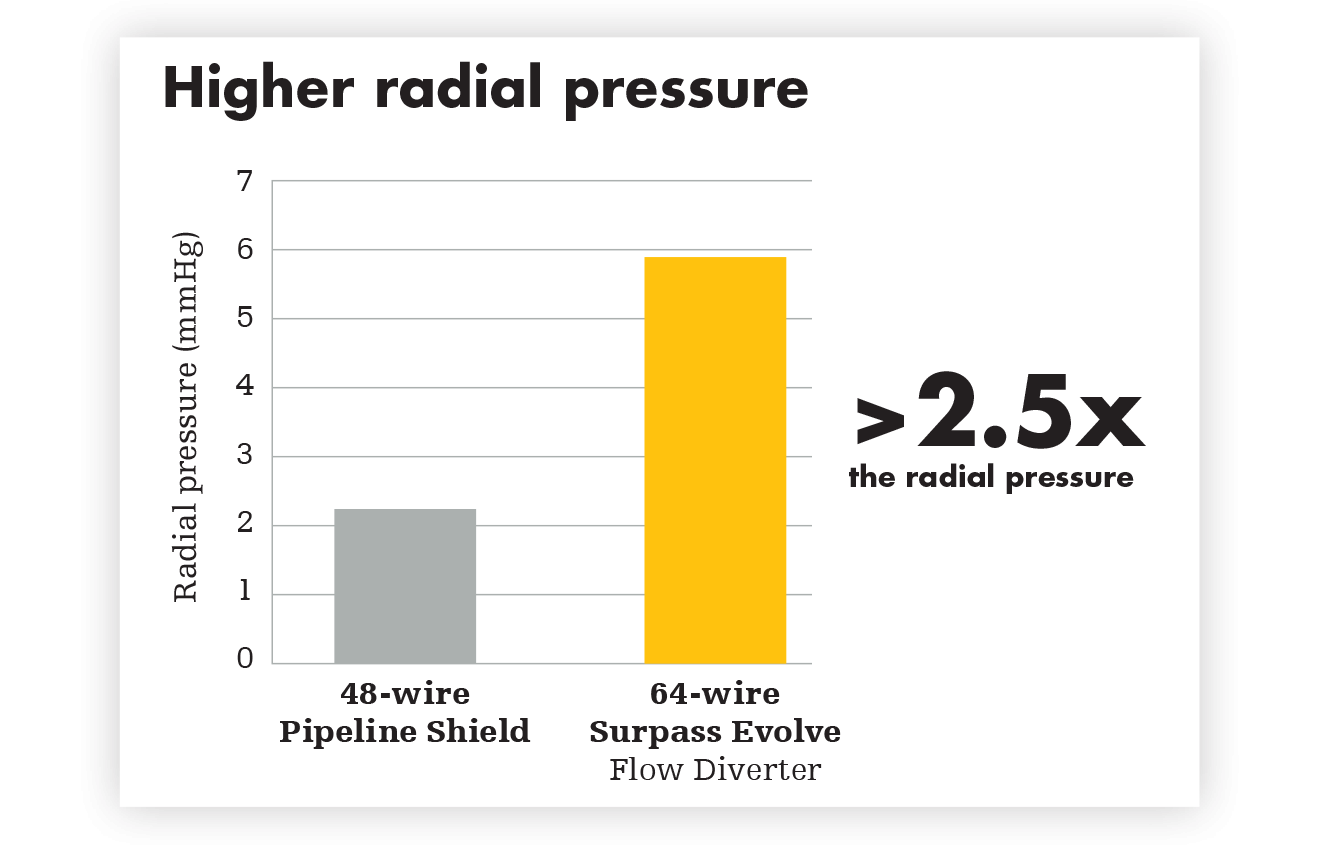
For reliable opening and complete apposition, Surpass Evolve Flow Diverter delivers. Ease of placement is a powerful advantage. With more wires generating higher radial pressure, Surpass Evolve Flow Diverter improves distal to proximal stent opening, while also increasing flexibility and conformability.
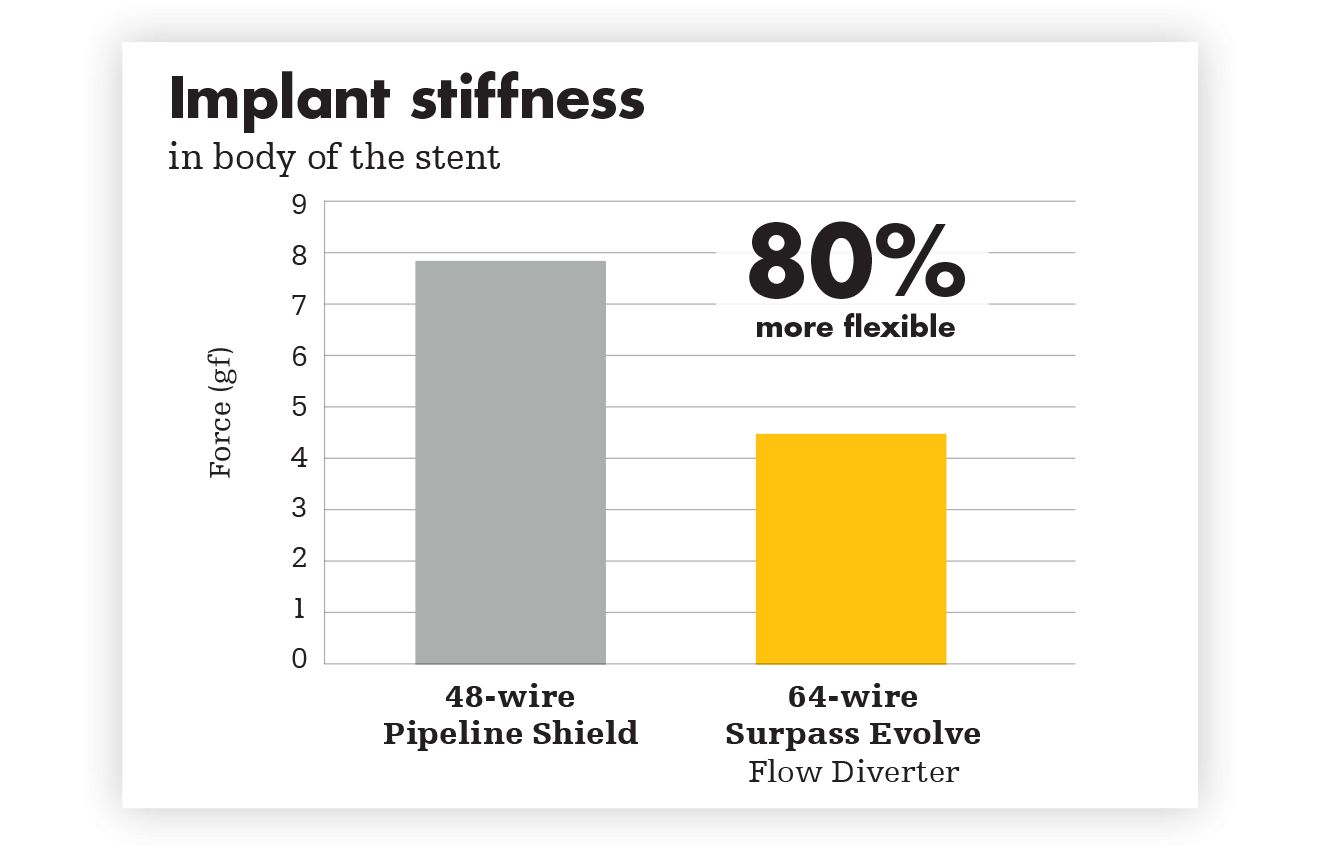
Testing performed by Stryker. Data on file at Stryker.
Bench test results may not necessarily be indicative of clinical performance.
Wired for diversion
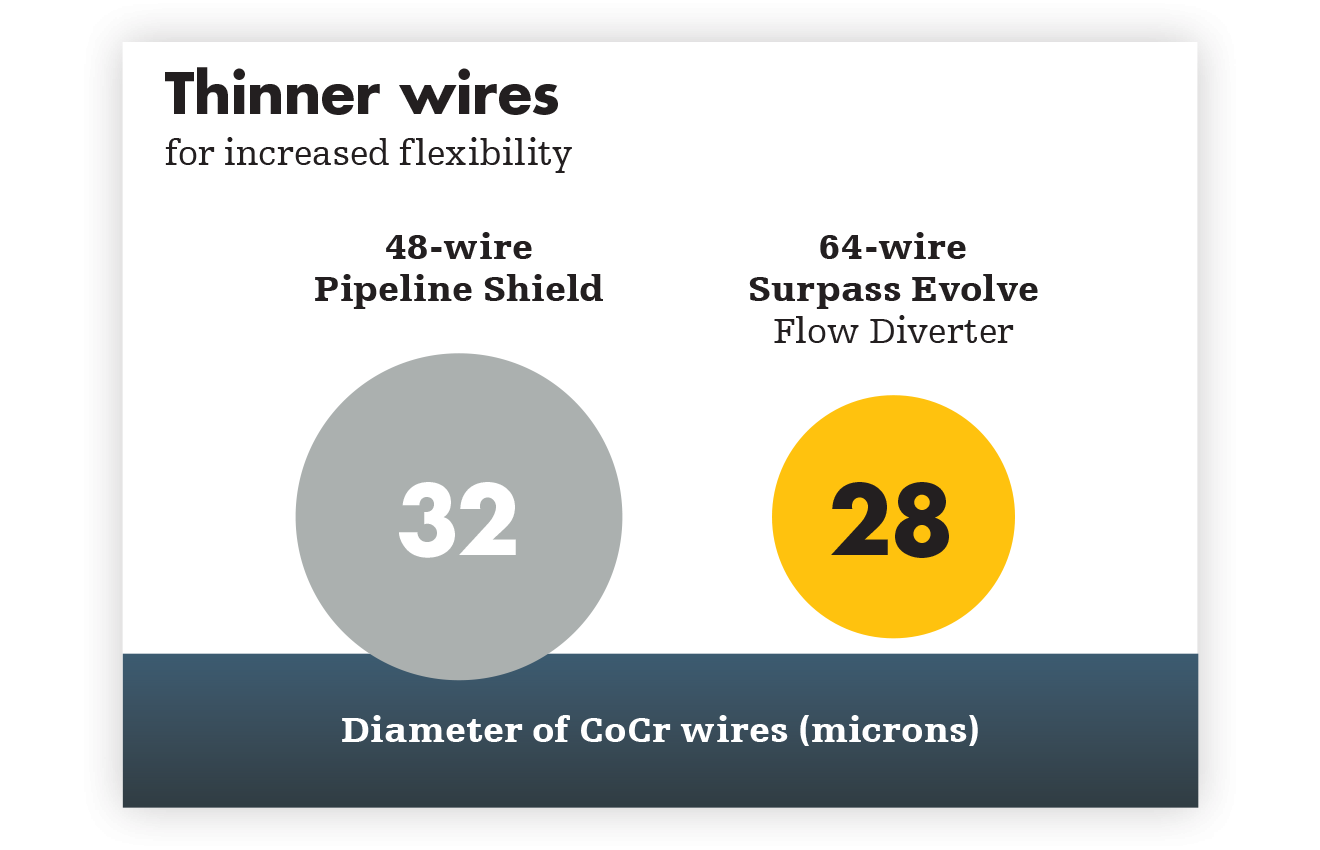
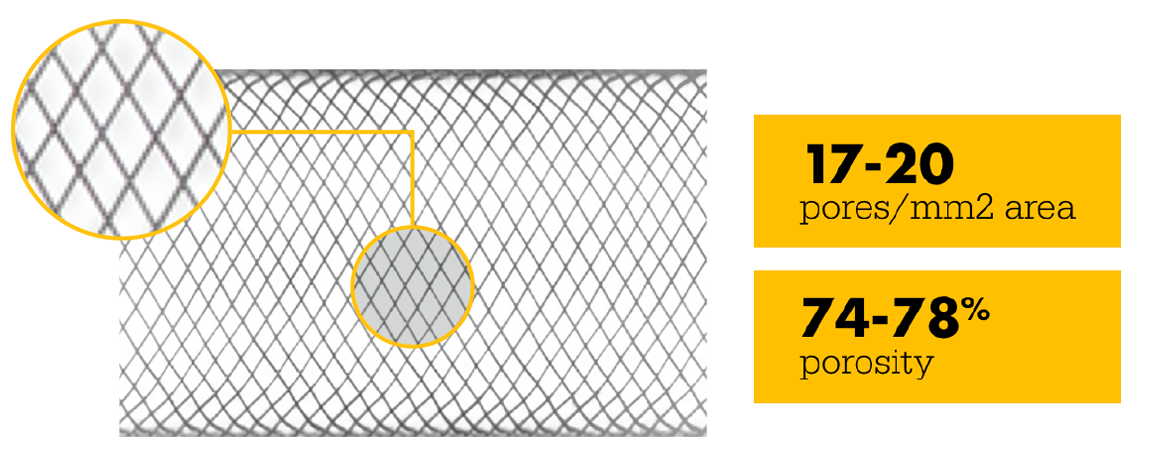
Higher mesh density creates higher flow diversion—and refines the standard. Get flow diversion efficacy in a single device. Designed with 64 thinner wires and a tailored braid angle, Surpass Evolve Flow Diverter gives you more flow diversion with the same porosity, across all sizes.
48-Wire Pipeline
Surpass Evolve Flow Diverter produced 30% more intraneurysmal flow stasis than the 48-wire Pipeline3

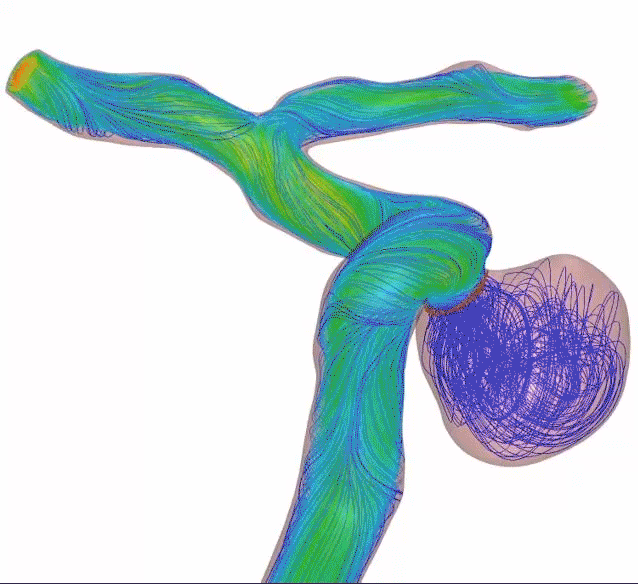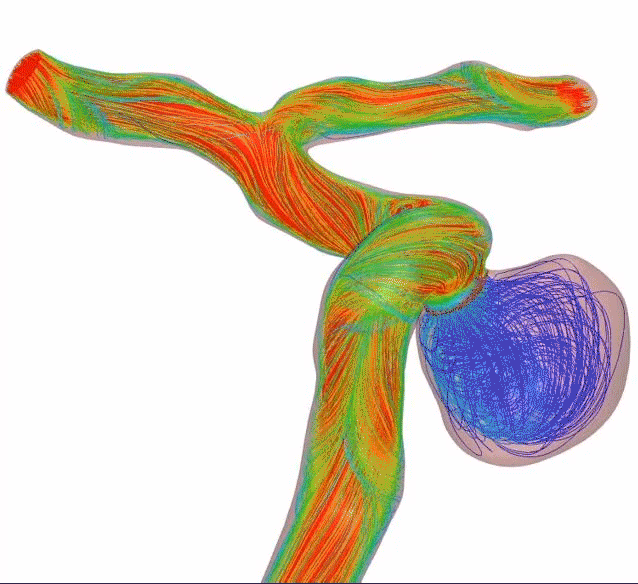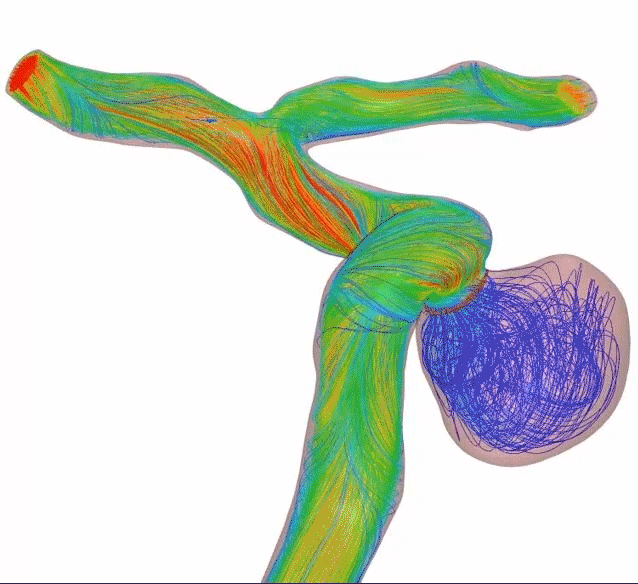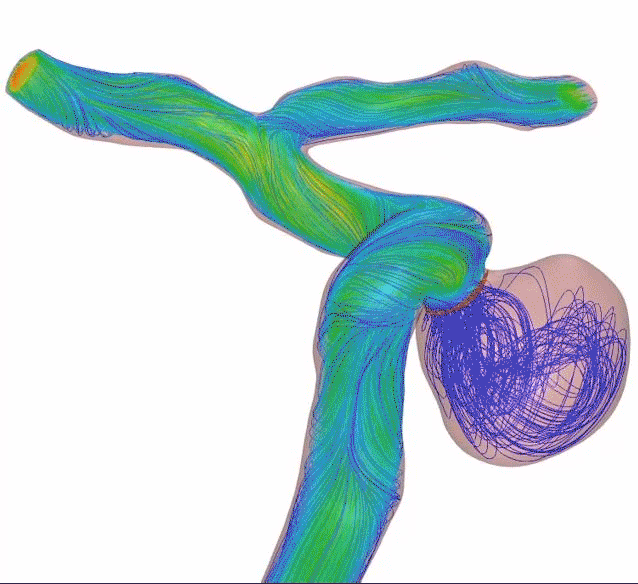
64-wire Surpass Evolve Flow Diverter
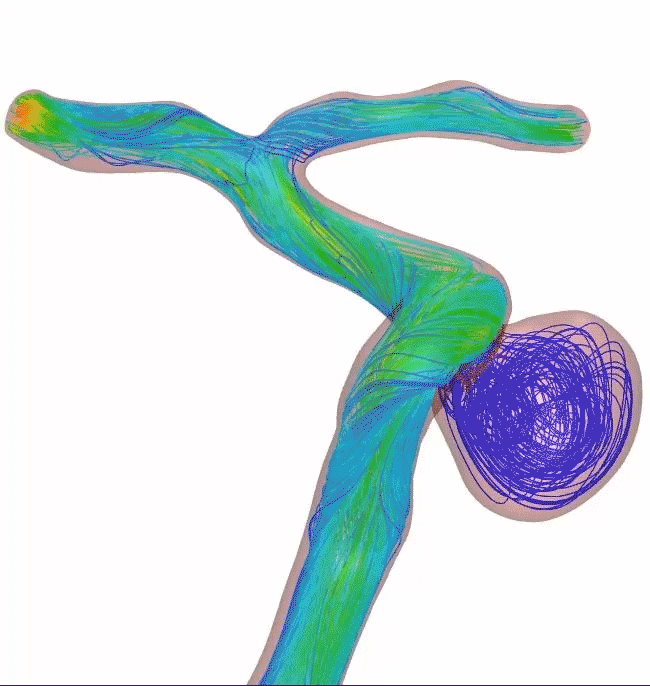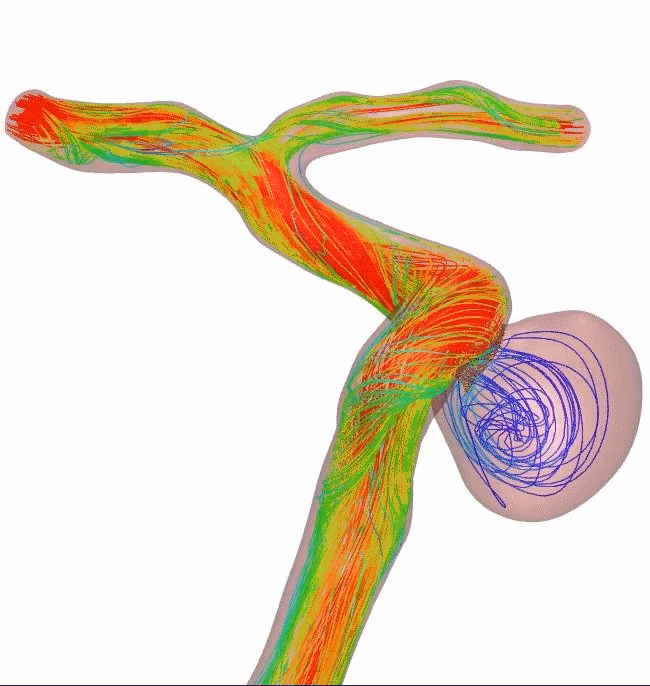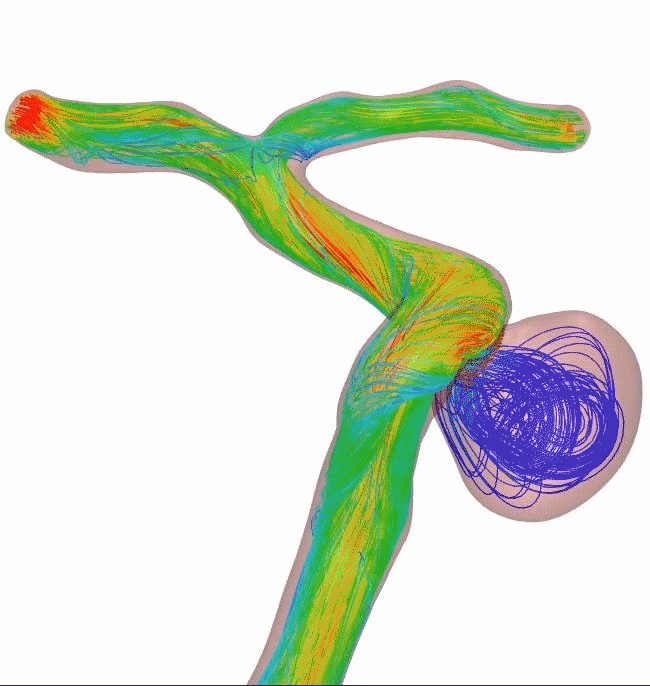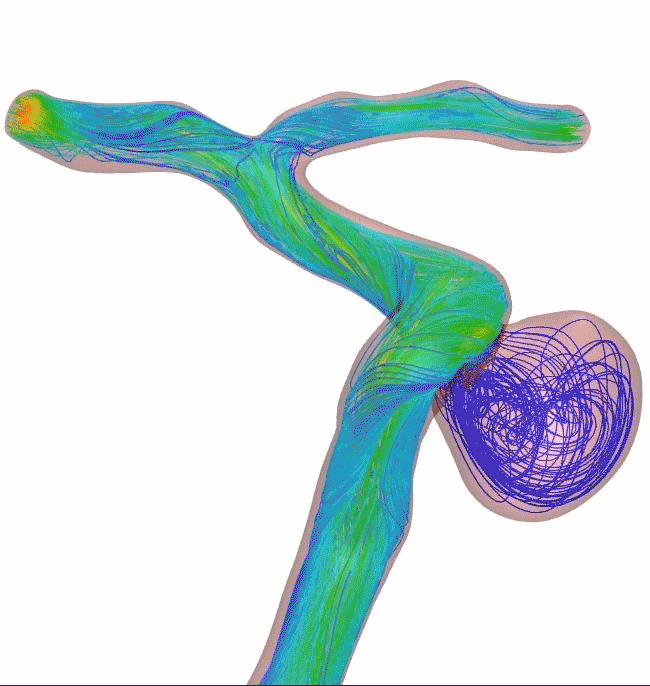
83% occlusion rate
<12mm aneurysms at 6 months with 1.2% complication rate2
Surpass Evolve® Flow Diverter in action
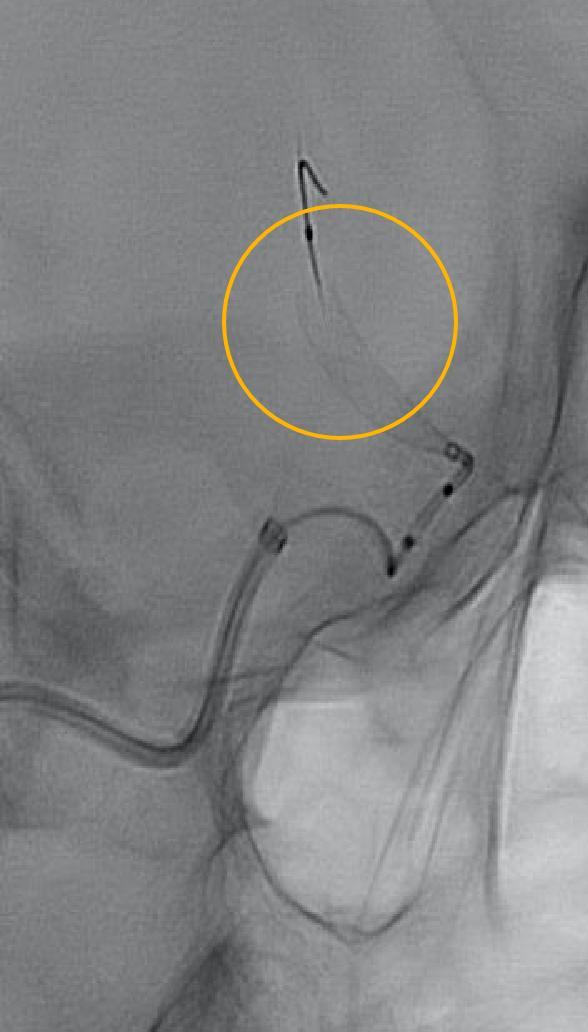
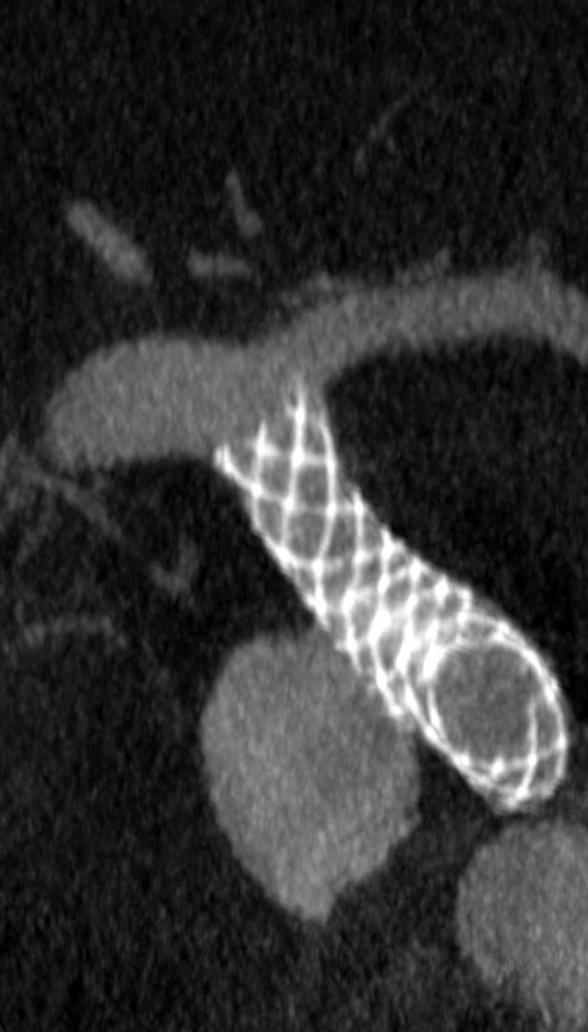
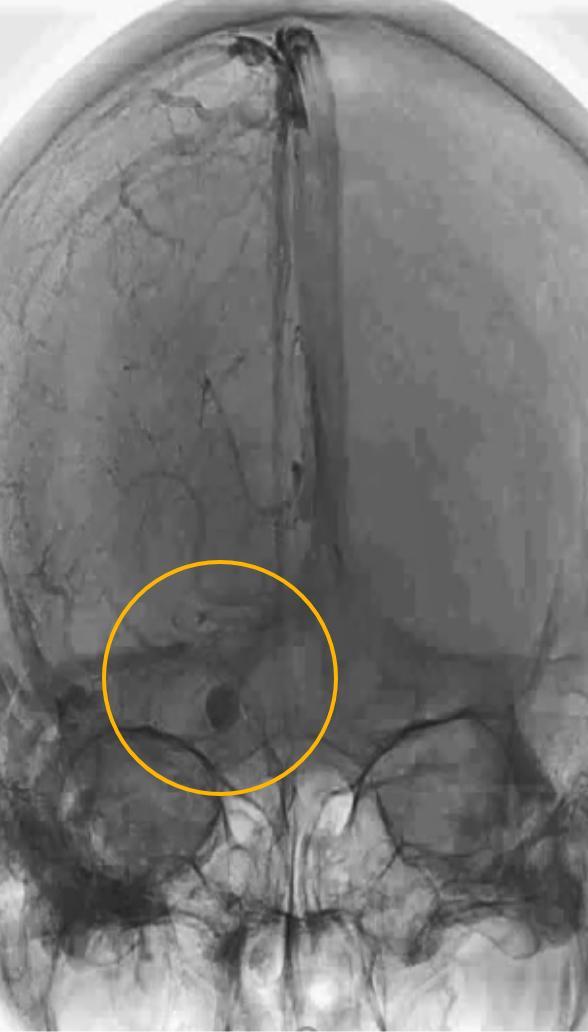
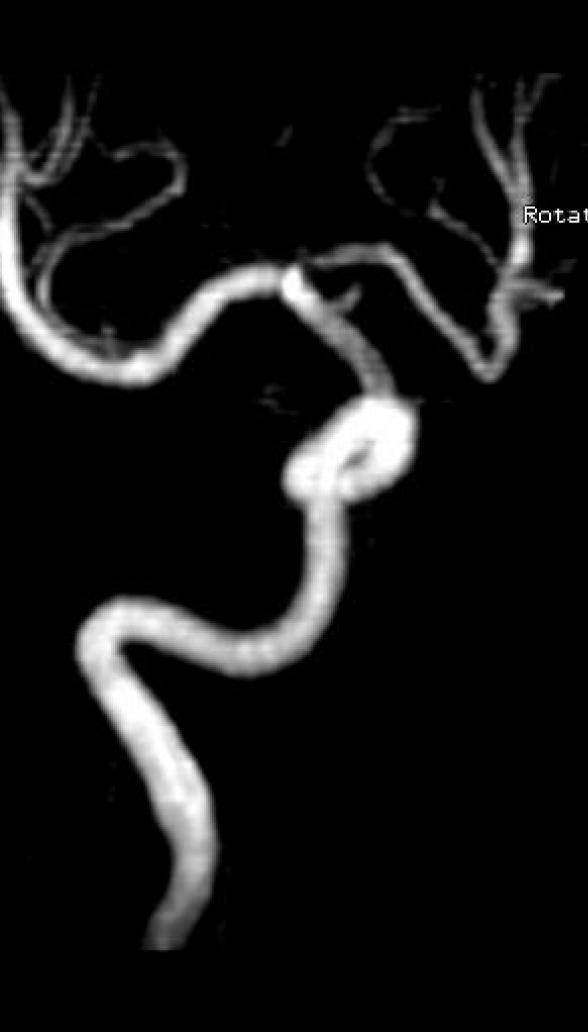
Supraclinoid aneurysm treated with Surpass Evolve Flow Diverter by Dr. Vitor Pereira at University of Toronto.
Results from case studies are not predictive of results in other cases. Results in other cases may vary.
1. Jee, T. K., Yeon, J. Y., Kim, K. H., Kim, J. S., Hong, S. C., & Jeon, P. (2021). Early clinical experience of using the Surpass Evolve flow diverter in the treatment of intracranial aneurysms. Neuroradiology, 1-9
2. LINNC Live Talk; November 2021, Orru et al. Long-Term Results for the Treatment of Intracranial Aneurysms with the Surpass Evolve Flow Diverter: a Multicenter Experience.
3. “Computational Fluid Dynamics Analysis to Compare Flow Diversion Efficacy of Evolve and Pipeline Devices”, SNIS 2020. Chander Sadasivan and Dr. David Fiorella
Important Safety Information
RX ONLY
See package insert for complete indications, contraindications, warnings and instructions for use.
Intended use / indications for use
The Surpass Evolve Flow Diverter System is indicated for
use in the endovascular treatment of patients (18 years of age and older) with unruptured large or giant saccular wide-neck (neck width ≥ 4 mm or dome-to-neck ratio < 2) or fusiform intracranial aneurysms in the internal carotid artery from the petrous segment to the terminus arising from a parent vessel with a diameter ≥ 2.5 mm and ≤ 5.0 mm.
Contraindications
- The Surpass Evolve Flow Diverter is contraindicated in the following patient types:
- Patients in whom the parent vessel size does not fall within
the indicated range. - Patients in whom antiplatelet and / or anticoagulation therapy
(e.g., aspirin and clopidogrel) is contraindicated. - Patients who have not received dual antiplatelet agents prior
to the procedure. - Patients with an active bacterial infection.
- Patients in whom the angiography demonstrates the anatomy
is not appropriate for endovascular treatment due to
conditions such as: - Severe intracranial vessel tortuosity or stenosis; and/or
- Intracranial vasospasm not responsive to medical therapy.
Potential adverse events
Risks that may be associated with the use of the Surpass Evolve Flow Diverter System in the intracranial arteries include:
- Adverse reaction to anesthesia, contrast or antiplatelet/
anticoagulation agents - Allergic reaction
- Aphasia
- Cardiac arrhythmia
- Cranial neuropathy
- Confusion, coma, change in mental status
- Death
- Device migration, fracture, misplacement
- Dissection of the parent artery
- Embolism (air, clots, device fragments)
- Groin injury (bleeding, pain, vessel/nerve damage) • Headache
- Hemiplegia
- Hydrocephalus
- Implant or parent vessel stenosis
- Implant thrombosis/occlusion
- Infection
- Intracerebral bleeding
- Mass effect
- Myocardial infarction
- Neurological deficits
- Perforation or rupture of aneurysm • Perforation of Parent Artery
- Progressive neurologic symptoms related to intracranial aneurysm (IA)
- Pseudoaneurysm formation
- Reaction to radiation exposure (i.e., alopecia, burns ranging in severity from skin reddening to ulcers, cataracts, delayed neoplasia)
- Renal failure
- Retroperitoneal hematoma
- Seizure
- Stroke
- Subarachnoid hemorrhage
- Thromboembolism from device
- Thrombosis of parent artery or branch vessel
- Transient ischemic attack (TIA)
- Vasospasm
Risks that are eye related with the use of the Surpass Evolve Flow Diverter System may include:
- Amaurosis fugax/transient blindness
- Blindness
- Diplopia
- Reduced visual acuity/field
- Retinal artery occlusion
- Retinal ischemia
- Retinal infarction
- Vision impairment
Warnings
- Contents supplied STERILE using an ethylene oxide (EO) process. Do not use if sterile barrier is damaged. If damage is found, call your Stryker Neurovascular representative.
- For single patient use only. Do not reuse, reprocess or resterilize. Reuse, reprocessing or resterilization may compromise the structural integrity of the device and/or lead to device failure which, in turn, may result in patient injury, illness or death. Reuse, reprocessing or resterilization may also create a risk of contamination of the device and/ or cause patient infection or cross-infection, including, but not limited to, the transmission of infectious disease(s) from one patient to another. Contamination of the device may lead to injury, illness or death of the patient.
- After use, dispose of product and packaging in accordance with hospital administrative and/ or local government policy.
- This device should only be used with adequate fluoroscopic guidance by physicians who have received appropriate training in interventional neuroradiology or interventional radiology, and training on the use of this device as established by Stryker Neurovascular.
- Persons allergic to nickel, cobalt chromium or platinum tungsten metal may suffer an allergic response to this system.
- Appropriate anti-platelet and anti-coagulation therapy should be employed in accordance with standard medical practice.
- [Clinical Warning] The safety and effectiveness of the device has not been evaluated or demonstrated for ruptured intracranial aneurysms.
- [Clinical Warning] A decrease in effectiveness has been observed in subjects aged > 65 years old, subjects with history of smoking and history of prior non-target intracranial aneurysm treated.
- [Clinical Warning] Placement of multiple Surpass Evolve Flow Diverters may increase the risk of ischemic complications. Implantation of more than one Surpass Evolve Flow Diverter in non-clinical in vivo studies showed an increase in the generation of platinum microparticulates in the surrounding vessel tissues. The presence of the platinum microparticulates did not produce any adverse pathological changes such as overt inflammation or altered healing in the implanted vessels in the animals; however, the risk of platinum microparticulates in human patients is unknown.
- [Clinical Warning] Delayed aneurysm rupture may occur with large and giant intracranial aneurysms.
Cautions / precautions
- Carefully weigh the benefits of treatment vs. the risks associated with treatment using the device for each individual patient based on their medical health status and risks factors for intracranial aneurysm rupture during their expected life time such as age, medical comorbidities, history of smoking, intracranial aneurysm size, location, and morphology, family history, history of prior asymptomatic subarachnoid hemorrhage (aSAH), documented growth of intracranial aneurysm on serial imaging, presence of multiple intracranial aneurysms, and presence of concurrent pathology. The benefits of device use may not outweigh the risks associated with the device in certain patients; therefore, judicious patient selection is recommended.
- This device has not been evaluated for pediatric use.
- The safety and effectiveness of the device has not been established in the treatment of small and medium wide-neck intracranial aneurysms.
- Experience with endovascular implants indicates that there is a risk of stenosis. Subsequent stenosis may require dilatation of the vessel segment containing the device. The risks and long-term outcome following dilatation of endothelialized devices is unknown at present.
- A thrombosing aneurysm may aggravate pre-existing or cause new symptoms of mass effect and may require medical therapy.
- Operators should take all necessary precautions to limit X-radiation dose to patients and themselves by using sufficient shielding, reducing fluoroscopy times, and modifying X-ray technical factors where possible.
- Lower intracranial aneurysm occlusion rates may be associated with giant intracranial aneurysms (>25mm).
- Lower intracranial aneurysm occlusion rates may be associated with implants that are not fully apposed to the vessel wall.
MRI safety information
Non-clinical testing demonstrated that the Surpass Evolve Flow Diverter is MR Conditional for single and overlapping up to 60mm in length. A patient with the Surpass Evolve Flow Diverter can be scanned safely in an MR system under the following conditions:
- Static magnetic field of 1.5 Tesla and 3 Tesla, only
- Maximum spatial gradient magnetic field of 3,000 Gauss/
cm (30 T/m) - Maximum MR system reported, whole body averaged specific
absorption rate (SAR) of 2 W/kg in the Normal Operating Mode Under the scan conditions defined, the Surpass Evolve Flow Diverter is expected to produce a maximum temperature rise
of 3.0°C after 15 minutes of continuous scanning (i.e., per pulse sequence). In non-clinical testing, the image artifact caused by the Surpass Evolve Flow Diverter extends approximately 10mm from this implant when imaged using a gradient echo pulse sequence and a 3 Tesla MR system. The lumen cannot be visualized on gradient echo or T1- weighted, spin echo pulse sequences. Caution: The Surpass Evolve Flow Diverter may create local field inhomogeneity and susceptibility artifacts during magnetic resonance angiography (MRA), which may degrade the diagnostic quality to assess effective intracranial aneurysm treatment. The health state of the patient or the presence of other implants may require reduction of the MRI limits
